Category: Uncategorized
Read coverage in EchoLive.ie on AVeta Medical with their VITA AV device set to help women suffering from vaginal atrophy
Great article by Deirdre McArdle for the EchoLive.ie as she continues her FemTech innovation series. This week AVeta Medical is featured along with founder Paula Newell. The device repairs vaginal…
Continue reading »Read Vish Gain’s article for the Silicon Republic on the release of the HIHI Telea pilot. “Pilot study of the Telea app gives hope to people with Parkinsons”
Read the full article here Pilot study of the Telea app gives hope to people with Parkinson’s
Continue reading »Great article on HIHI’s pilot with Telea in the Irish Examiner – UCC start-up hopes to improve speech for people with Parkinson’s disease through new app
Read Michelle McGlynn’s article here https://www.irishexaminer.com/news/munster/arid-41371588.html
Continue reading »Great coverage in Irish Tech News on HIHI’s product pilot with Telea which saw a 10% increase in voice in patients with Parkinson’s Disease
Read the full article and promising results of the pilot here. https://irishtechnews.ie/world-parkinsons-day-telea-offers-new-hope/
Continue reading »
On World Parkinson’s Day HIHI and Telea release results of new pilot study which offers hope to people living with Parkinsons disease
Irish pilot study sparks new hope for people with Parkinson’s disease, prolonging their ability to communicate by slowing down the progression of the disease on vocal function. A HIHI pilot…
Continue reading »Read about Saor Health with their app which will help women feel safer exercising outside. The app is a winning product from the HIHI FemTech Call competition
Read Deirdre McArdle’s full article in WOW (Women on Wednesday) on the Saor Health’s app. It is part of a FemTech series spotlighting the winners of the FemTech Innovation Call…
Continue reading »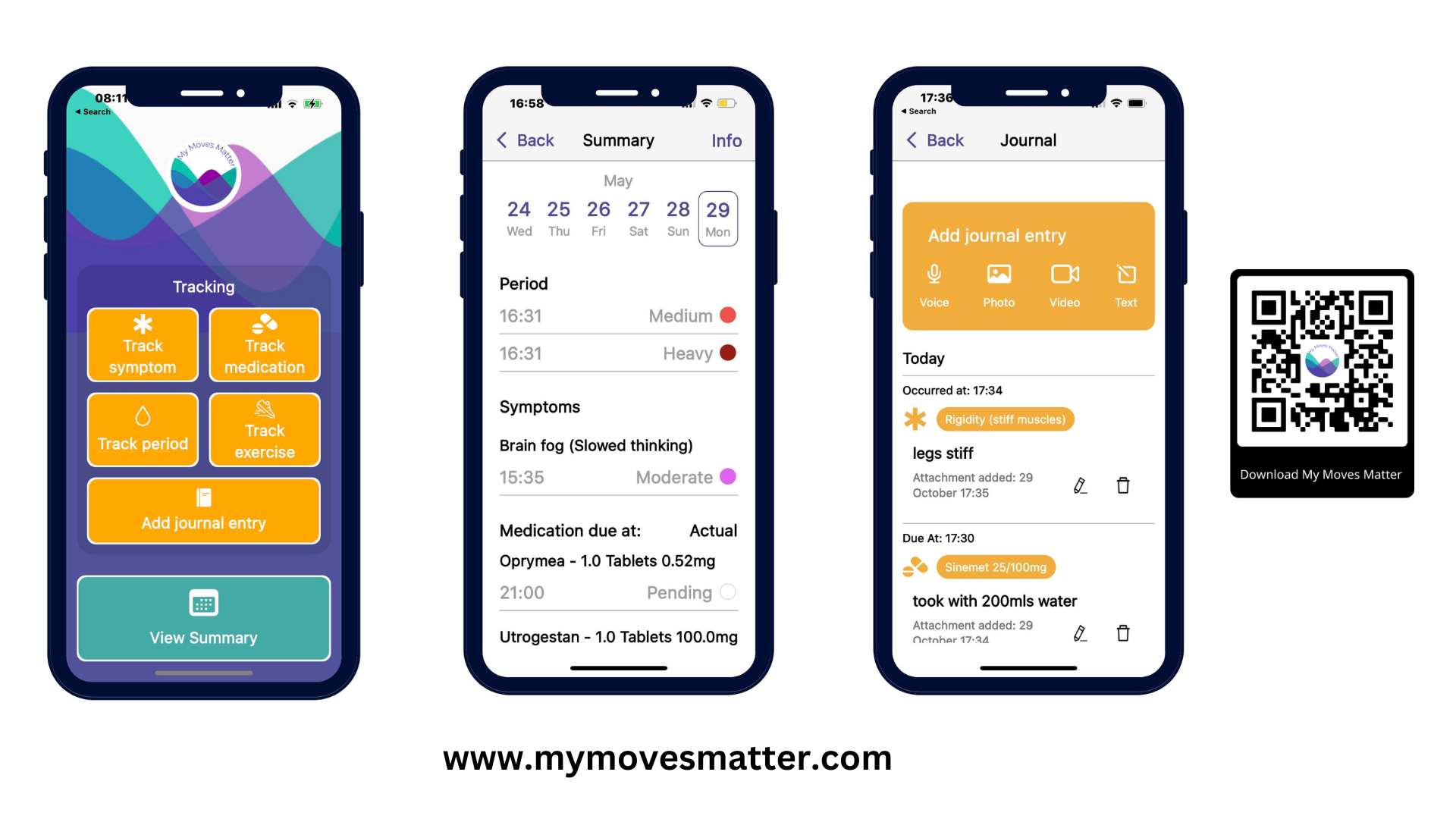
Read the feature in WOW this week on My Moves Matter an app designed to help Parkinsons patients track their symptoms
Read Deirdre McArdle’s full article with founder of My Moves Matter Richelle Flanagan, a winner of HIHI’s FemTech call competition. https://www.echolive.ie/wow/arid-41362345.html
Continue reading »
The HIHI Team were delighted to present at the recent WHO meeting in Copenhagen “Building a Public Health Innovation Ecosystem”
The Health Innovation Hub Ireland team were delighted to present recently in Copenhagen at the WHO Regional Office for Europe meeting “Building a Public Health Innovation Ecosystem”. Our colleagues Dr…
Continue reading »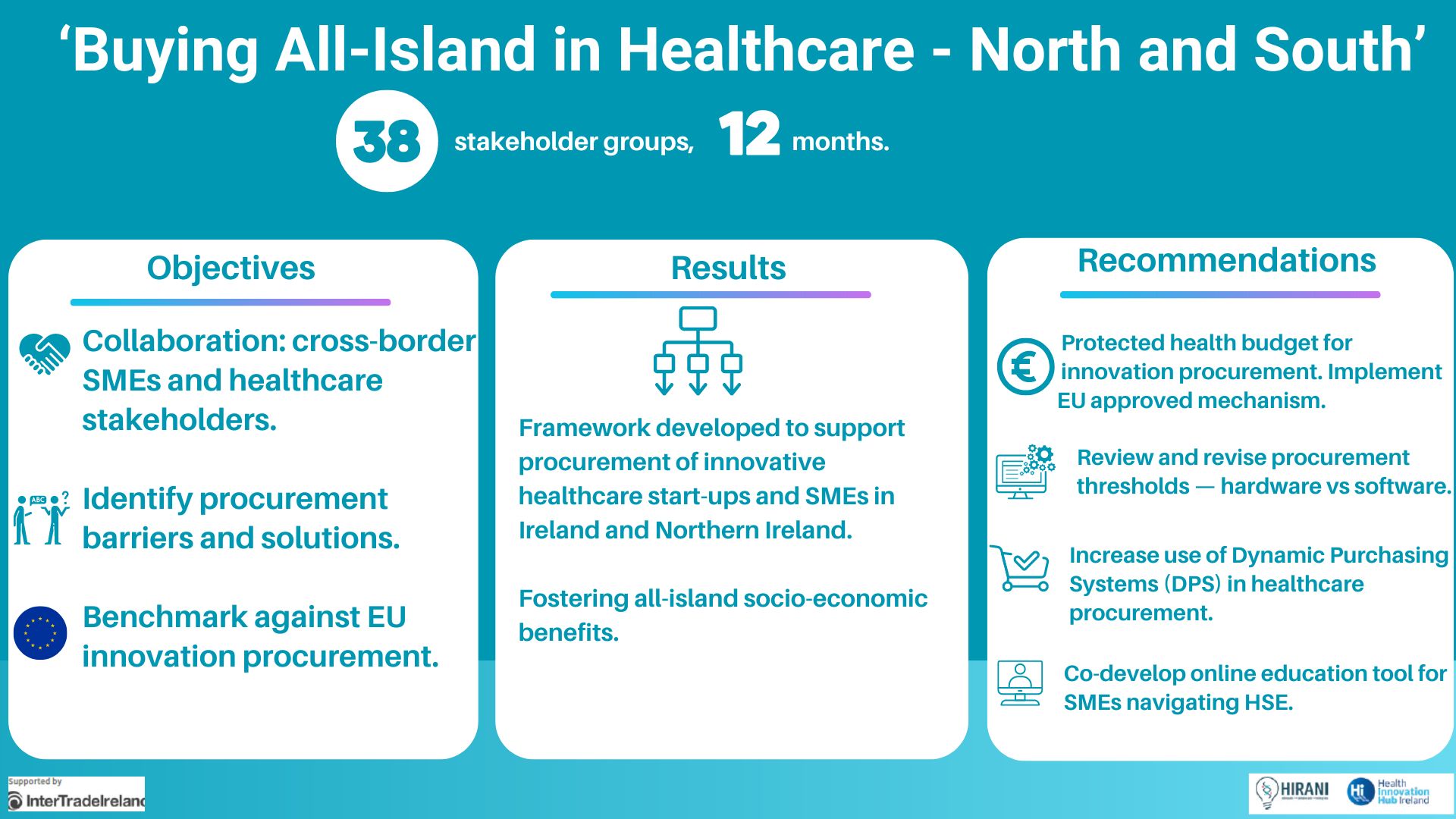
Newstalk Breakfast covers findings of new HIHI report
The ‘Buying All-Island in Healthcare – North and South’ report was launched yesterday with an interview on Newstalk Breakfast. This was carried across all national bulletins throughout the day and…
Continue reading »
New HIHI cross-border report finds common barriers for smaller vendors in healthcare and urges an all-island shift to innovation procurement, enabling SMEs and Start-ups to thrive in domestic health tech markets
Launched today, the ‘Buying All-Island in Healthcare – North and South’ report publishes the findings and recommendations framework of the All-Island Medtech SMEs (AIMS) initiative, delivered through a 12-month cross-border…
Continue reading »
Smart D8 Call for pilots Community Health, Health Literacy and Environment – Closes this Friday 29
Smart D8 Opens New Call for Pilot Projects following three years of success New call for pilot projects in three areas: Community Health, Health Literacy and Environment Up to six…
Continue reading »Read an article in this weeks’ WOW, Women on Wednesdays featuring identifyHer, a HIHI supported company, with their new tech sensor which monitors the menopause
Read Deirdre McArdle article for WOW (Women on Wednesday) for the Evening Echo featuring HIHI supported company identifyHer. New tech is getting to the crux of perimenopause (echolive.ie) identifyHer was…
Continue reading »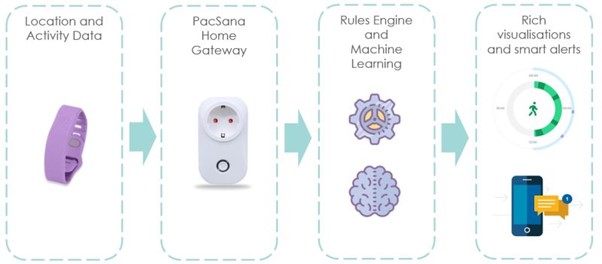
Gamechanging wearable sensor technology set to prolong independent living for Ireland’s elderly population
HIHI is this week spotlighting a game changing wearable technology for elderly people living in the community in Ireland. As the Irish population over the age of 65 increases over…
Continue reading »Vox-AI amongst winners of HIHI’s FemTech Innovation Call with an AI driven tool centred on menopause management
Vox-AI is an AI-driven platform that uses advanced Retrieval Augmented Generation (RAG) technology, complemented by multilingual spoken word bots and avatars. MenoMate is a chatbot centred on menopause management and…
Continue reading »In advance of International Women’s Health Week – Meet one of the winners of HIHI’s FemTech Innovation Call indentifyHer
identifyHer Ltd is a femtech company focussed on detecting and tracking menopausal symptoms. The identifyHer platform is a wearable biosensor to detect and track menopausal symptoms with an accompanying application to present…
Continue reading »Dr Tanya Mulcahy Director of HIHI and Founder of Femtech Ireland to speak at the upcoming National Menopause Summit – Advocating for change
HIHI are delighted to announce that Dr Tanya Mulcahy will speak again this year at the National Menopause Summit. The event takes place on the 11th and 12th April 2024…
Continue reading »The Menopause Hub among the winning companies of HIHI’s FemTech Innovation competition
The Menopause Hub App will allow women to track their menopause and perimenopause symptoms prior and post treatment and to book appointments. It will provide medication and appointment reminders and has…
Continue reading »Meet Aurin another winning company from HIHI’s FemTech Innovation competition that aims to relieve the symptoms of menopause
The AURIN platform is a femtech innovation for the treatment of menopausal vasomotor symptoms (hot flushes and night sweats). AURIN comes from a team of researchers from University of Galway and University…
Continue reading »A Postpartum product from PPR Healthcare among winners of HIHI Femtech Innovation competition
PPR Healthcare develop products to help the recovery process after childbirth for mothers. Mul tie Briefs is a new product range in development from PPR Healthcare. Read about the product which…
Continue reading »Meet HIHI’s FemTech Innovation Call winners. Spotlighting OnaWave Medical with a product Endosolve set to be a gamechanger for the diagnosis and treatment of pelvic conditions
Congratulations to OnaWave Medical on being one of the 11 winning companies emerging from HIHI’s first Femtech Innovation Call competition. A medtech company, it focusses on improving the diagnosis and…
Continue reading »








